PiVR has been developed by David Tadres and Matthieu Louis (Louis Lab).
7. Tools
Important
The software has different functionality if run on a Raspberry Pi as compared to any other PC. This manual is for the PC (Windows, MacOS and Linux if not run on a Raspberry Pi) version of the software
7.4. Display tracked experiment
This tool allows you to display the tracked animal in its arena similar to a video player.
The experiment must have been generated using PiVR: either on on the Raspberry Pi using the Tracking or Virtual reality or by using the post-hoc analysis option in the ‘Tools’ Menu.
Besides enabling you to conveniently see where the animal was in each frame, this tool allows you to:
Correct false Head/Tail assignments by swapping them
Save a video (in mp4 format) of the experiment.
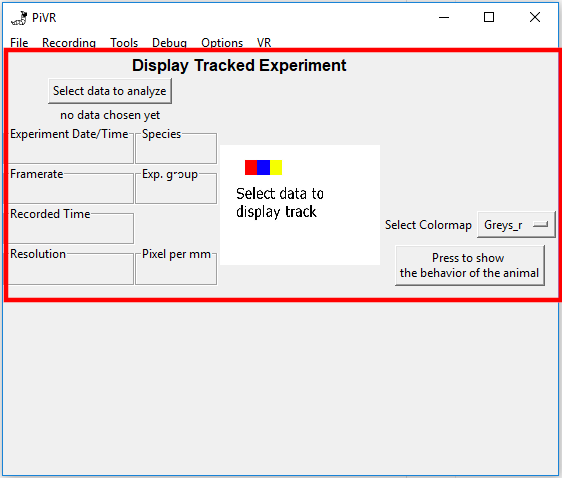
To display an experiment, press on the “Select data to analyze” button and select a folder containing a single experiment.
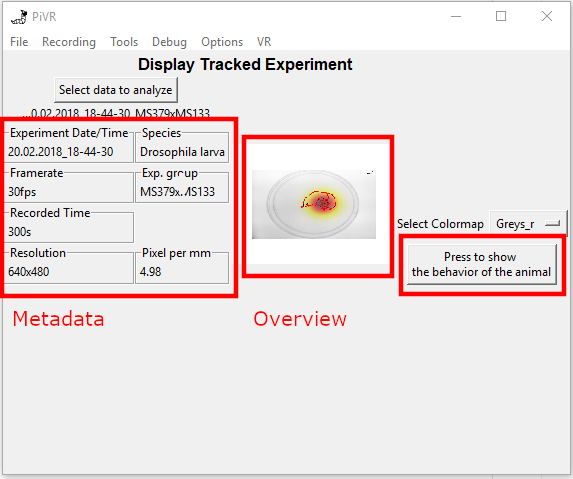
The software will automatically read the metadata of the experiment and will be displayed on the left side of the GUI.
In the center, the ‘Overview of tracking.png’ file is shown.
On the right you may choose a colormap before pressing “Press to show behavior of the animal”.
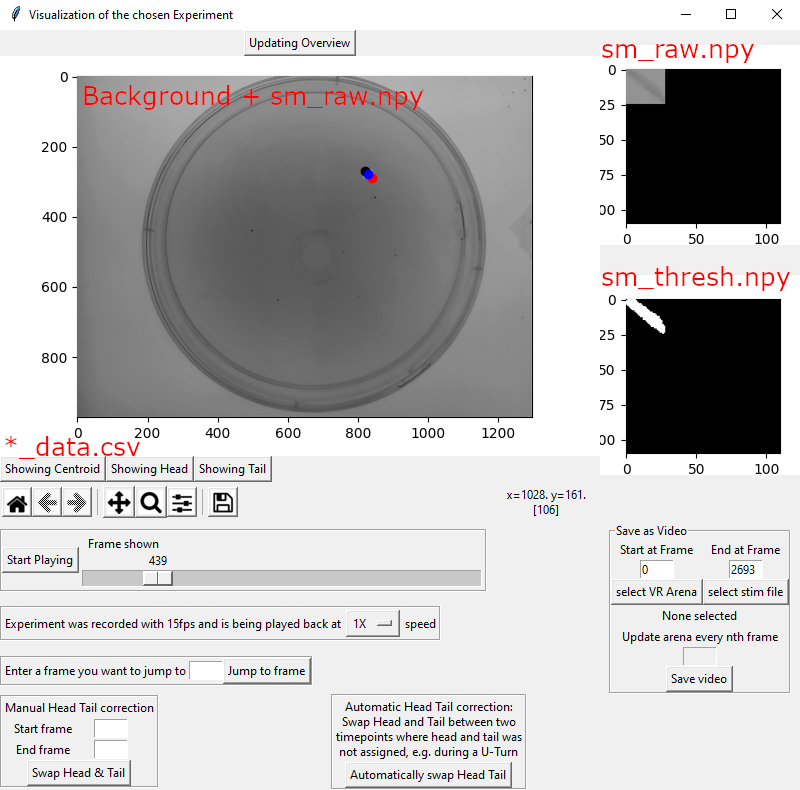
The window above will emerge as a popup. While it is open the main GUI is unavailable!
This window has the following functionality:
Updating Overview: This button will allow you to turn the main window off when playing back the experiment. This can be useful if you are only interested in the small images on the right.
The main figure in the center of the window is created by placing the raw small image (sm_raw.npy) into the reconstructed background image (Background.jpg) using the bounding box coordinates (bounding_boxes.npy). It also displays the detected Centroid, Tail and Head (*_data.csv) directly on the animal.
The three buttons below the main window on the left, “Showing Centroid”, “Showing Head” and “Showing Tail” allow you to turn off the different parts shown in the main figure.
The toolbar below allows you to interact with the main window (zoom etc.).
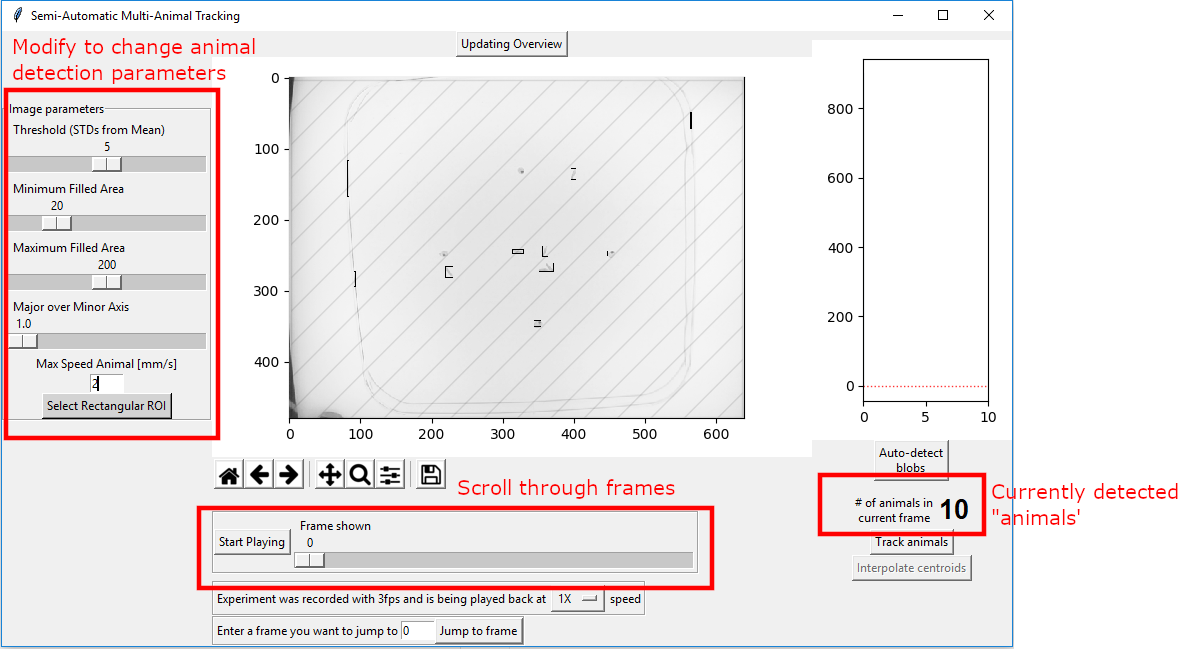
The slider below lets you scroll through the experiment. Pressing the “Start Playing” button will display the experiment.
The Dropdown menu below the slider lets you play back the experiment at a variety of speeds you can choose from.
If there is a particular frame you want to go to, you can enter that number in the field below and press “Jump to frame”.
If the head/tail assignment has been made incorrectly, you have two options to fix it here.
Manual Head Tail correction: Define the timepoints in which the head and tail have been incorrectly identified in ‘Start frame’ and ‘End frame. Press ‘Swap Head & Tail’
Automatic Head Tail correction: You need to slide the slider to a frame with incorrect head tail classification. Then press ‘Automatically swap Head Tail’. The algorithm will swap head and tail for previous and following frames until it finds a frame where the head and tail could not be assigned, for example during a U-Turn.
On the top right, the small raw image (sm_raw.npy) is being displayed.
Below, the binary small image (sm_thresh.npy) is being displayed.
7.4.1. Creation of a video file
In the “Save as Video” box at the bottom right you may create a video of the experiment. There are several options to customize the output file:
Note
If you get an error message it is possible that you have not installed the correct version of ffmpeg. Usually the error message should indicate what you have to install. If you need help, please open a ticket.
If you want to create a video displaying the the animal at the correct position, all you need to do is define the start and end frame of the video and hit ‘Save Video’. If you do not want the head, tail and/or centroid to be displayed in the video, turn it off as you would for the interactive visualization.
If you have been stimulating your animal with a time dependent stimulus and you want your video to indicate when the stimulus was turned on, press the button titled ‘select stim file’ and select your stimulus file. The stimulus file must adhere to the PiVR guidlines. Then hit ‘Save Video’
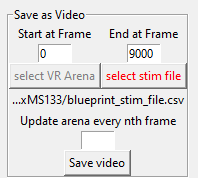
If you have been stimulating your animal with a static Virtual Arena and you want your video to indicate where the animal was relative to the position of the Virtual Arena. Press the button titled ‘select VR arena’ and select your VR file. Then hit ‘Save Video’
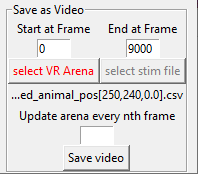
If you have been presenting your animal with a dynamic virtual reality (i.e. one that changes over time) you may also include this in the video. Press the button titled ‘select VR arena’ and select the file you used when presenting the animal with the dynamic virtual reality. Finally, indicate in ‘Update arena every nth frame’ after how many frames the arena needs to be updated. For example, if you have been presenting an arena that updates at 15Hz while recording at 30Hz you need to update every 2nd frame (30/15=2).
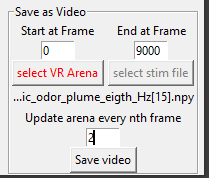
When pressing “Save video” the video will be saved as “Video.mp4” in the experimental folder. This usually takes a significant amount of time, even on a fast computer.
7.5. Multi-Animal Tracking
This tool allows you to identify more than one animal in a video or in a series of images.
The identification of the animals is achieved via background subtraction of the mean image. Each animal is given an arbitrary number. In the next frame, the animal closest to that number in the previous frame is assigned.
This guide is intended to get you started quickly. If you are
interested in the more technical aspects, please see
multi_animal_tracking.MultiAnimalTracking
Note
Identifying more than one animal in an experiment is computationally challenging. There are several specialized tools such as the multiworm tracker, Ctrax, idtracker, idtracker.ai and MARGO. The PiVR multi-animal tracking software has not been benchmarked against these tools. This software has several limitations. It is probably ok to use in cases where you are interested in counting how many animals are in a general area. It is not recommend to use the tracker for other parameters, such as calculating animal speed (due to loss of identity after collision and ‘jumps’ in the trajectory) and similar parameters.
After selecting a folder containing a video of image sequence of an experiment containing multiple animals, press the “Press to show the behavior of the animals” button.
The software will now load the image data (which can take a considerable amount of time) and display a new window which will help you to optimize the tracking parameters.
Note
You might notice that the image is distorted. This is due to the Raspberry Pi camera lens. See here.
First, have a look at the all-important “# of animals in the current frame” on the bottom right of the popup. In this video there are only 6 animals, but the algorithm detects 10 objects as “animals”. The blobs identified as animals are indicated using small rectangles in the main figure.
Important
The goal is to have have as many frames as possible contain only the expected amount of animals.
To achieve this goal, start by defining the area in which animals can move into. In this particular experiment, the outline of the petri dish can be clearly seen:
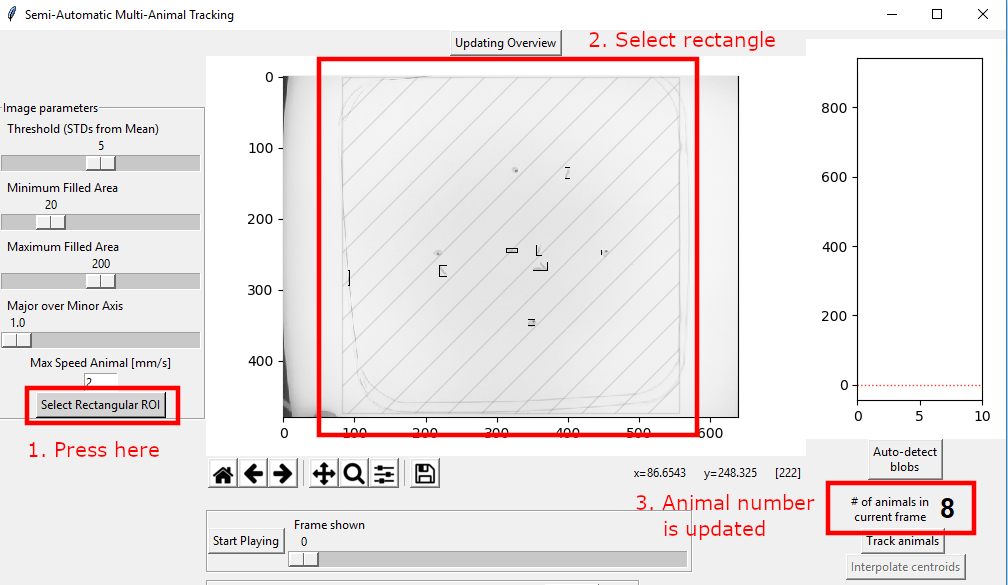
Press the “Select Rectangular ROI”. The button will turn red.
Using the mouse, create a rectangle in the main window. The result is indicated. The “# of animals in the current frame” is immediately updated.
While this is already better, there are still two blobs wrongly identified as animals, one at (y=300,x=100) and the other at (y=270, x=480).
By increasing the “Threshold (STD from Mean) number these mis-identified blobs are not mistaken for animals animal:
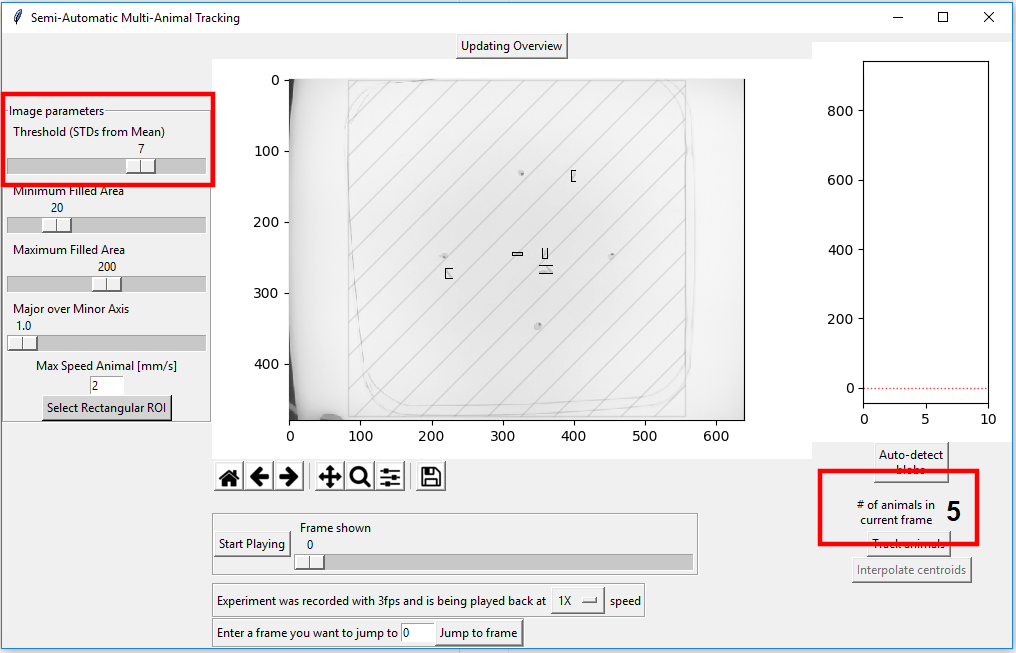
There are now no mis-identified animals. While there are a total of 6 animals in the image, the algorithm can only detect 5. To understand why that is the case, press the magnifying class symbol below the main figure and draw a rectangle around the region you want to enlarge.
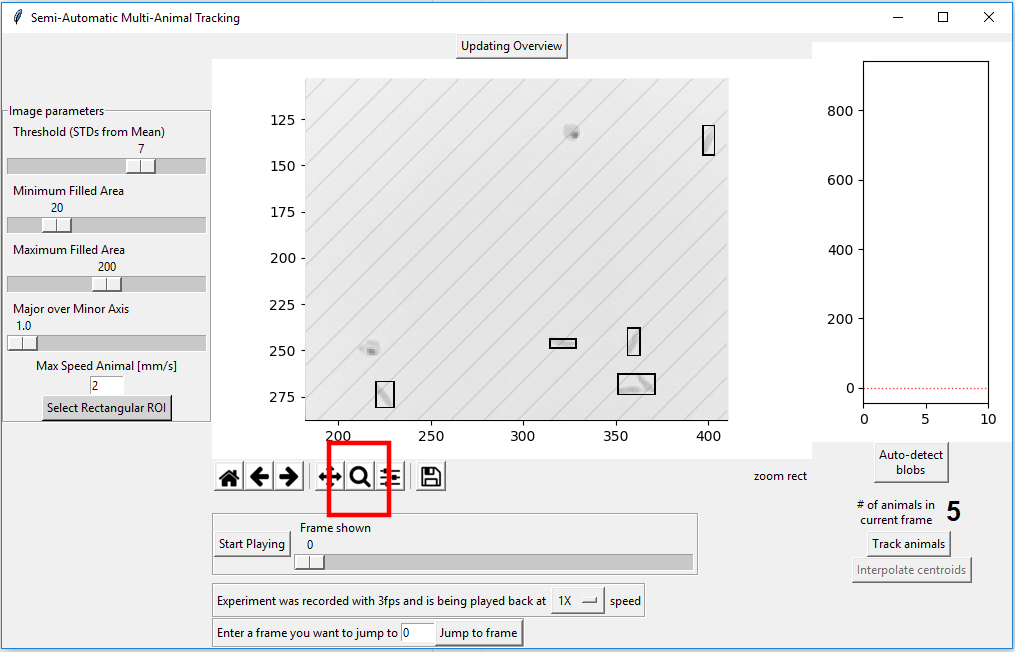
It is now obvious that 4 out of 6 animals are properly identified. Two of the animals, at (y=260, x=360) are very close to each other and are identified as one animals, however. There is no way for this tracking algorithm to separate animals that are touching each other!
Now you need to make sure that the image parameters are such that you get the expected animal number in all frames. To not have to go through each of the frames manually, you can just press “Auto-detect blobs” on the bottom right. This will run a script that will take the current image detection parameters into account and just count how many blobs are counted as animals. The result is plotted in the figure on the top right.
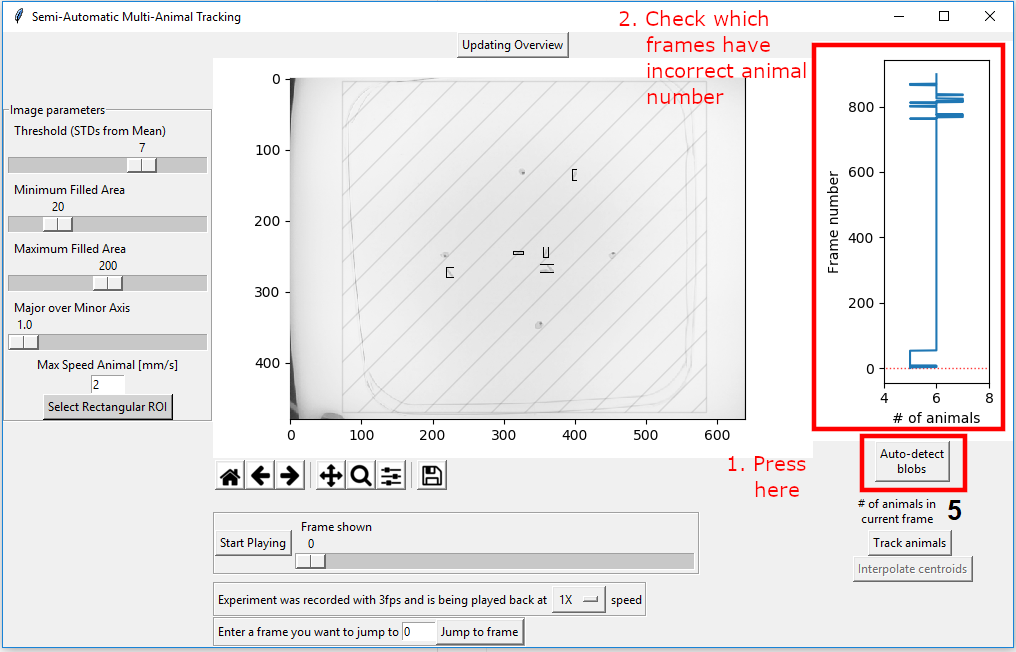
This result indicates that at the beginning of the video there are several frames where only 5 animals can be detected. By visually inspecting these frames it becomes obvious that this is due to the two animals touching each other as they already do in the first frame.
Next, find frames that have the wrong amount of animals. Try to fix them using the image parameter settings.
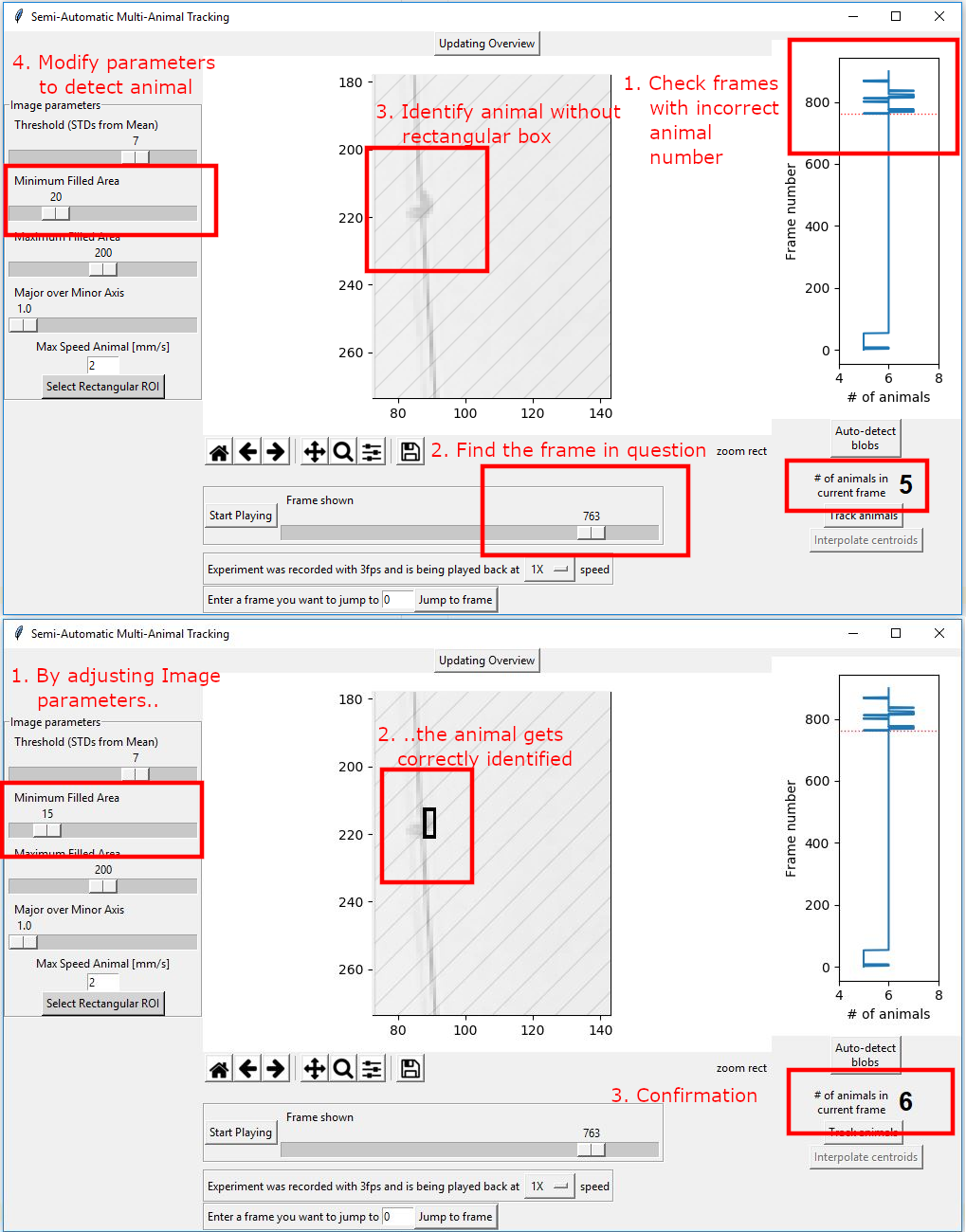
There can be situations where the animal number will just be wrong and it can not be fixed. The algorithm can handle this if the number of those frames is low.
Once you have optimized the image parameters, go to the frame where the correct amount of animals is detected. This is crucial as it tell the algorithm how many animals to expect!. Then press “Track animals”.
A new popup will open. It indicates how many animals (and where) are detected in this frame. Each animal gets an arbitrary number. If the number of animals is correct, press “Looks good, start tracking”. Else press “Not good, take me back”.
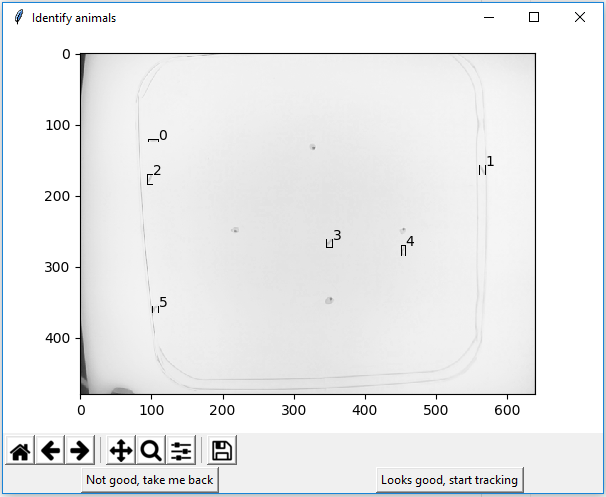
The tracking itself is computationally quite expensive and therefore usually takes a while to complete. To speed up tracking, you can press the “Updating Overview” button above the main window.
Once tracking has concluded the result will be displayed in the main window as shown below.
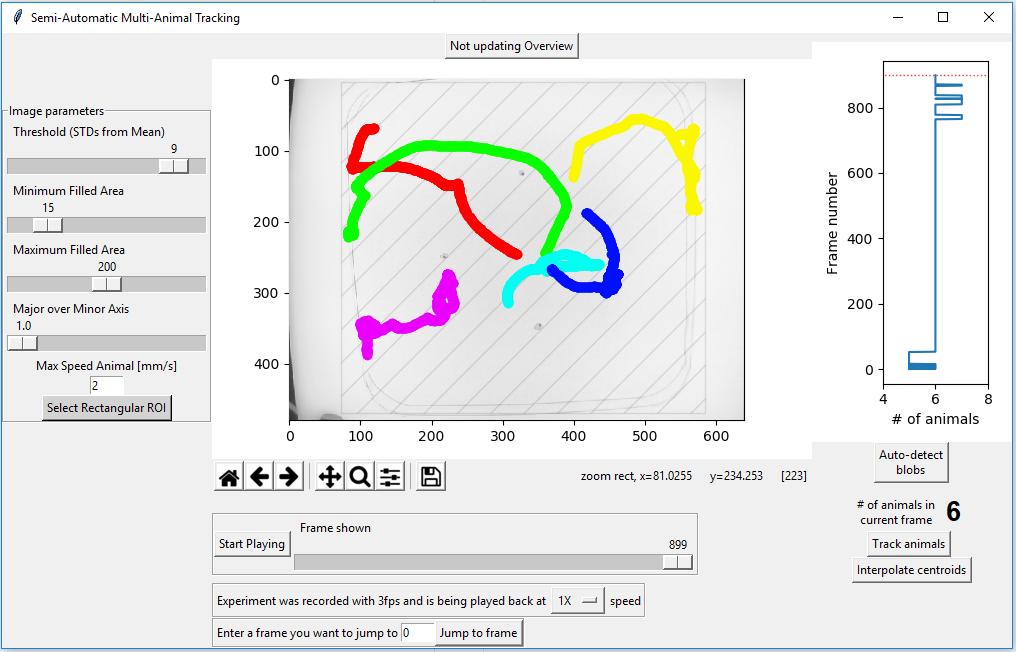
If there are huge gaps in the trajectory, for example because an animal could not be detected for a while, you can press the “interpolate centroids” button. This will calculate realistic possible distances travelled (based on ‘Max Speed Animal[mm/s]) between frames and try to connect trajectories. This is a untested feature - use at your own risk. Ideally you should not have to use this option.
7.5.1. Multi Animal Tracking Output
After using the Multi-Animal Tracker, you will find two new files in the experimental folder:
The “Background.npy” file which is just the mean of all images, resulting in the background image used during tracking.
The “XY_positions.csv” file contains the X and Y coordinates for each identified animal for each frame. For frames with not enough animals, the corresponding row will be empty.
7.6. Creating a new Virtual Arena
How to create a new virtual arena, the essential tool that gives PiVR its name?
First, a brief overview of what the different elements of an arena file mean:
You can find several example virtual arena files in the PiVR/VR_arenas folder: As an example lets analyze “640x480_checkerboard.csv”
The file itself is a 2D matrix with 640 columns and 480 rows. Each value in the matrix defines what happens on that pixel on the camera.
For example, if you define the value 100 at position column = 75 and row = 90 here in the virtual arena and then present this virtual arena to the animal using the Closed Loop stimulation tab on PiVR, if the animal is at pixel column = 75 and row = 90 the intensity of 100 will be played back.
A big advantage of virtual realities over real environments is the control the experimenter has over the experimental conditions. When running an experiment, one often has to repeat trials many times. In real environments, the experimenter can never have identical initial conditions, e.g. the animals is placed at a slightly different position, the animal moves in different conditions before the tracking even starts etc. With virtual reality, this factor (which often introduces variability into data) can be alleviated. The experimenter can define a virtual reality arena and the animal will always be presented with the identical initial conditions.
To do this, lets examine another example virtual arena file you can find in PiVR/VR_arenas: “640x480_gaussian_centred_animal_pos[250, 240,0.0].csv”
This file has a string at the end of its filename: animal_pos[250, 240,0.0]. This string indicates where the animal must start in relation to the virtual reality. In this example, wherever the animal is in the real image when the experiment starts, the virtual reality will be translated so that it is at x-coordinate 250 and y coordinate 240.
In addition, if the third value (here 0.0) is defined, the movement of the animal during detection is taken into account: The virtual arena is rotated so that the animal always starts going into the same direction relative to the virtual arena. The angle you may use goes from -pi to +pi (see atan2).
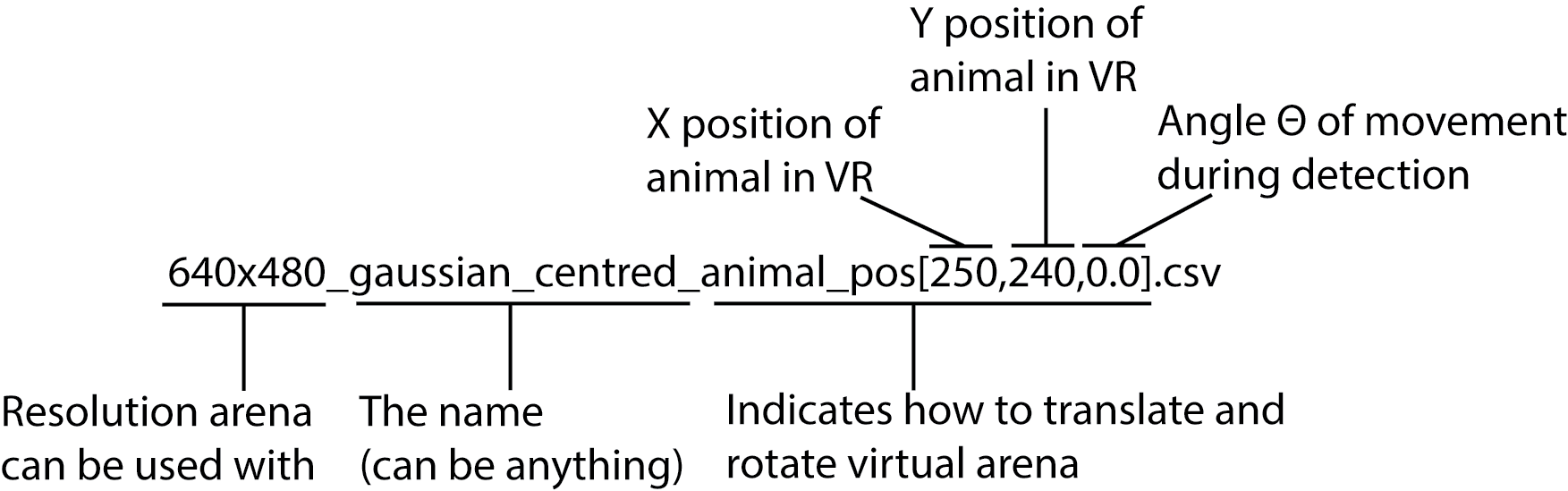
If you want to create a virtual reality arena from scratch, for example in python, all you need to do is create a matrix with the correct dimension, fill it with values between 0 and 100 as you see fit and export the file as csv.
import numpy as np
virtual_arena = np.zeros((480,640),dtype=np.float64)
# Define parts of the arena where the animal is supposed to be
# stimulated e.g. by typing
virtual_arena[:,0:100] = 100
virtual_arena[:,101:200] = 75
virtual_arena[:,201:300] = 50
# this will give you a very coarse grained virtual arena that will
# stimulate strongly if the animal is on the left side, stimulate
# 75% if the animal is a bit more on the right and 50% if it is
# still on the left but almost in the middle. The rest is
# unstimulated as of yet.
# Now you need to save the arena. Let say you want to have the animal
# start ascending the gradient from the middle (essentially animal
# has to move to the left in the virtual arena)
np.savetext("Path/On/Your/PC/640x480_my_awesome_arena_animal_pos[300,240,0.0].csv")
Alternatively, you can use the “Tools->Draw VR Arena” option on the PC version of PiVR.
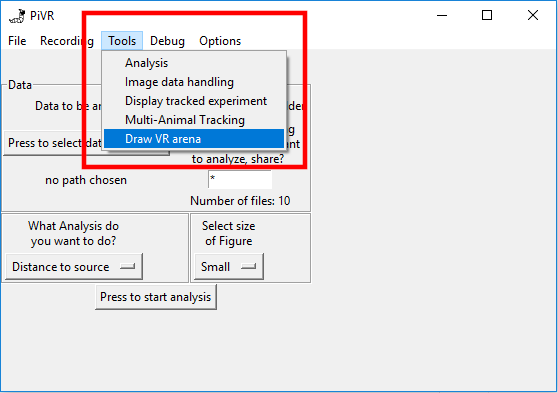
You will find the following empty canvas. You can open a previously defined virtual arena or work on the blank canvas. Either way, you have the the option to create gaussian shaped 2D circles and rectangles. To “draw” such a gaussian shaped 2D circle, you can either press on “Draw Gaussian Circle with Mouse” (and then click somewhere on the canvas) or you can press on “Draw Gaussian Circle at defined coordinates”.
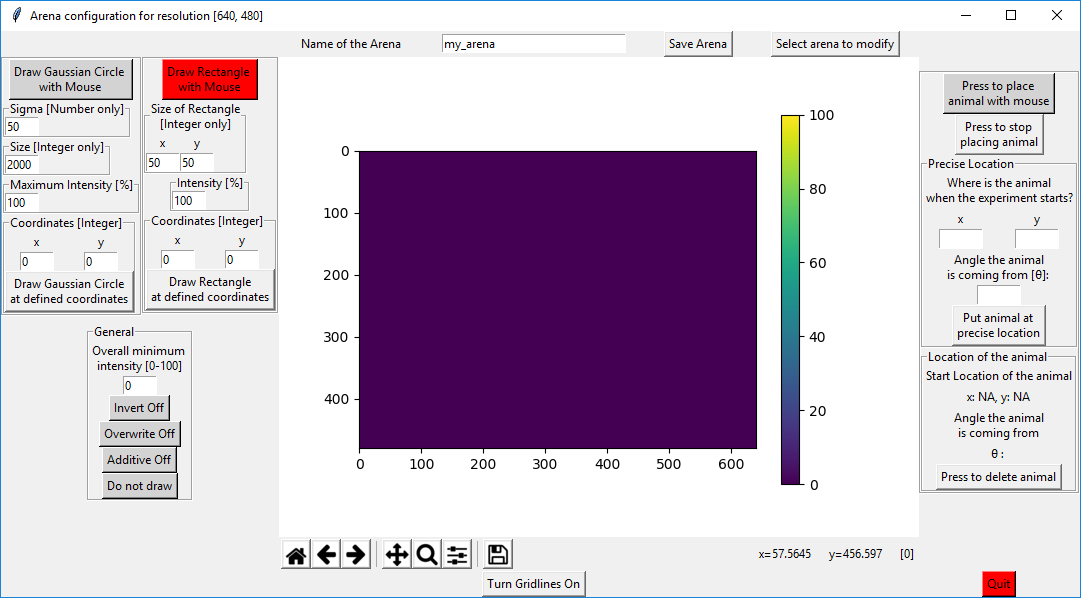
You can change the Gaussian shaped 2D circle by changing its Sigma, its size and the intensity.
Analogous, you can define the size and the intensity of the rectangle by entering the desired value.
In the example below, I used the standard settings for the gaussian circle size but changed the “Coordinates” to the values shown. Then I pressed on “Draw Gaussian Circle at defined coordinates”(red squares). Then I modified the “Coordinates” of the rectangle on the right to x=100 and y=100 and pressed on “Draw Rectangle at defined coordinates” (green squares).
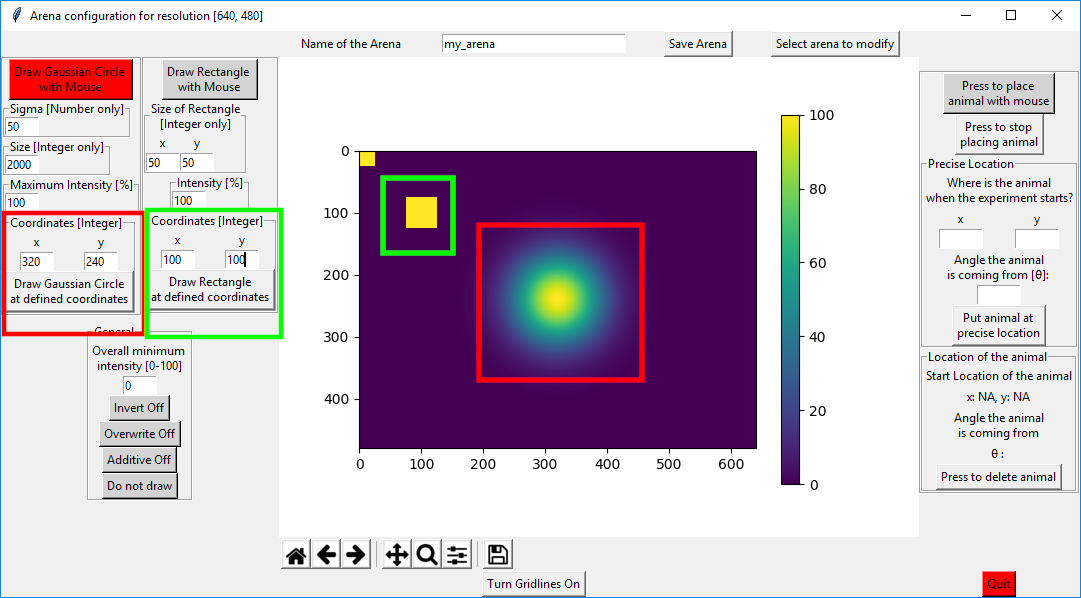
There are 3 additional buttons that expand the possibilities of drawing virtual arenas:
Invert: If this is on, whenever you draw a circle or rectangle, it subtracts the values from the intensity that is already present. This option was used to create the following virtual arena: “640x480_volcano_animal_pos[320,240,0.0].csv”
Overwrite: will just overwrite the previous pixel values with the new values with no regard to the previous value (as opposed to “Invert” and “Additive”)
Additive: If you place a 50% rectangle somewhere and then place another on top of it, usually nothing will happen as the absolute value is being drawn. If this is on, the values are “added”.
Note
If you go above 100%, everything will be normalized.
On the right side of the canvas you can define the starting position of the animal in the virtual arena. Besides just x and y position, you can define the direction from which the animal is coming from.
You can use the mouse, either by clicking (just x and y position) or by clicking and dragging (x, y and angle). Alternatively, you can use precise coordinates.
Note
Angle can go from -pi to +pi. See atan2 for visualization.
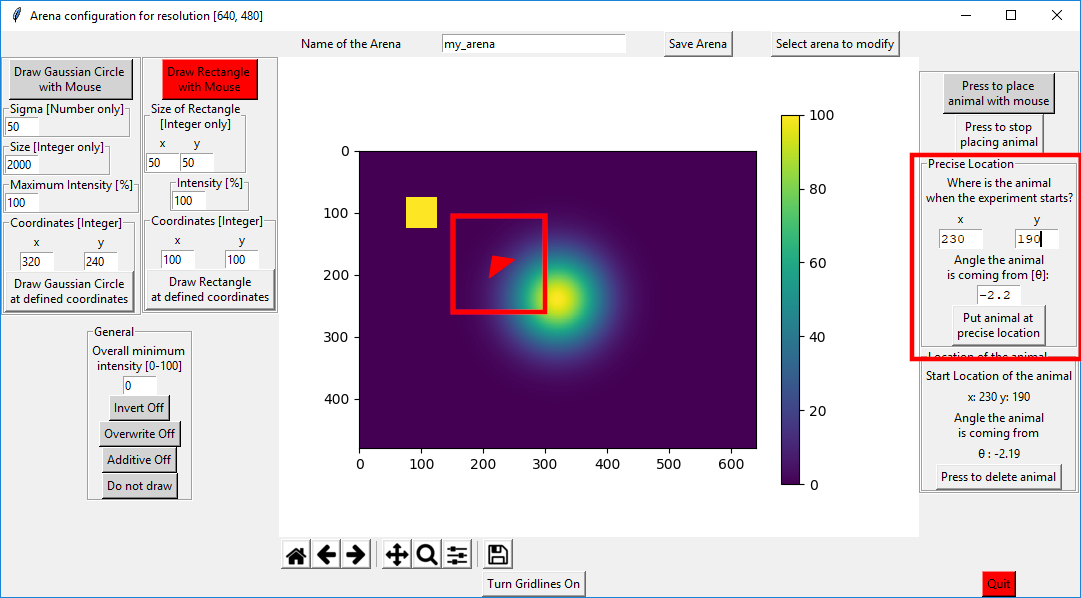
Once you are done with the arena, make sure to save it. Then you can just quit the window.
7.7. Creating a new Dynamic Virtual Reality
Before attempting to create a new dynamic virtual reality please make sure you understand exactly how a static virtual reality is created.
The main differences between the static and the virtual reality are:
Usage of numpy arrays instead of csv files.
The filename includes the speed at which the virtual reality is “played back” as a “Hz[#]” term.
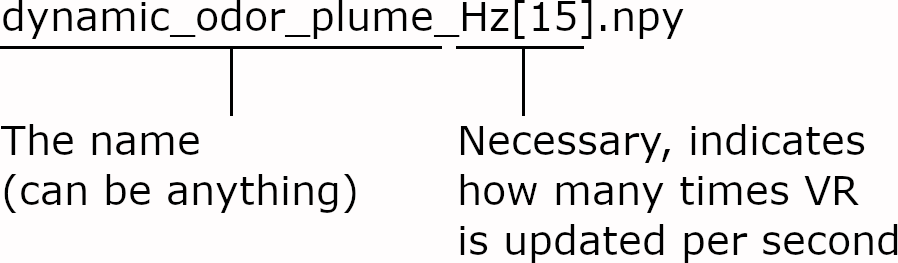
Instead of going from 0 - 100, stimulation in defined as 8 bit (0 - 255)
For details on how exactly this is implemented, please see here
There are several limitations compared to static virtual realities:
Dynamic virtual realities can not orient themselves relative to the the position of the animal at the start of the experiment.
RAM of PiVR is limited (but this is getting better with the RPi4 which has up to 4Gb). As the arena that is presented must be loaded into RAM at the beginning of the experiment, it can not be too large. On our RPi3+ (1Gb of RAM) we successfully used dynamic virtual realities of 135Mb for 5 minute experiments.
It is not possible to use the “Adjust Power” field!
How does a dynamic virtual reality file look like? Essentially it is just the 3D version of the static virtual arena with the difference that an integer value of 255 indicates that the LED is completely on (as opposed to 100 in the static case). The third axis contains all the ‘frames’ of the virtual reality ‘video’.
As an example, follow the instructions below to convert a real odor plume measurement to a dynamic virtual reality that PiVR can use:
Get the odor plume data from Álvarez-Salvado et al ., 2018 and unzip the file. The relevant file is “10302017_10cms_bounded_2.h5”.
While downloading, you can make sure you have the correct conda environment to handle the data. Below is the code to create new environment and install the necessary packages (Windows):
conda create -n dynamic_odor_env python=3.7 -y activate dynamic_odor_env conda install -c anaconda h5py -y conda install matplotlib -y
Now start python and adapt the following code:
import numpy as np import h5py # Adapt this to the location of your file data_path = 'C:\\10302017_10cms_bounded_2.h5' # Read the h5 datafile using the h5py library data = h5py.File(data_path, 'r') # The h5 datafile is a dictionary. As the keys are unkown, # cycle through to learn what files are stored in this fie. for key in data.keys(): print(key) #Names of the groups in HDF5 file. # ok, the relevant (only) key is 'dataset2' # Using this group key we can now access the data actual_data_temp = data['dataset2'] actual_data = actual_data[()] # make sure you do have the data: print(actual_data.shape) # (3600, 406, 216) # This information, together with the metadate indicates that # the first dimension holds the measurements of the timepoints # (Frequency of 15Hz for a duration of 4 minutes = 3600 frames). # The other two dimensions are spatial dimensions. # Below the reason it is necessary to use uint8 (8bit integer # precision instead of float (just remove the #) # import sys # print('Using 64bit float precision: ' + # repr(sys.getsizeof(np.zeros((actual_data.shape[0],640,480), # dtype=np.float64))/1e6) + 'Megabytes' ) # print('Better to use uint8 as I ll only need to use: ' + # repr(sys.getsizeof(np.zeros((actual_data.shape[0],640, 480), # dtype=np.uint8))/1e6) + 'Megabytes in RAM' ) # Now to convert the 64bit floating point to 8 bit # find maximum value: max_value = np.amax(actual_data) # find minimum value: min_value = np.amin(actual_data) # convert data to uint8 to save a ton of space convert_factor = 255/(max_value + (- min_value)) downsampled = (actual_data+(-min_value)) * convert_factor # We need to create an empty arena with 640x480 pixels using the # correct datatype.. correct_size_arena = np.zeros((480,640,actual_data.shape[0]), dtype=np.uint8) # and place the moving arena in to the center. start_y = int(correct_size_arena.shape[0]/2 - downsampled.T.shape[0]/2) # 132.0 start_x = int((correct_size_arena.shape[1]/2 - downsampled.T.shape[1]/2)) # 117 correct_size_arena[start_y:int(start_y+downsampled.T.shape[0]), start_x:int(start_x+downsampled.T.shape[1]),:] = downsampled.T # In order to see how the just created arena looks like, run # the code below (uncomment by removeing the #) # import matplotlib.pyplot as plt # fig,ax = plt.subplots() # ax.imshow(correct_size_arena[:, :, 0]) # plt.show() # Even after downsampling, file is way to large (>1Gb). To # solve this, only save 1/8 of the file (~30seconds). np.save('C:\\dynamic_odor_plume_eigth_Hz[15].npy', correct_size_arena[:,:,0:int(correct_size_arena.shape[2]/8)])
Now you must transfer the file containing the new dynamic virtual reality to your PiVR setup and select it when running your experiment, identical to what you would do to select a static virtual arena.